|
The results of the analyses, measured by low-field 19F TD-NMR, supported the reported label claims for the average drug content.īased on the simplicity and reproducibility of the analysis, we envision this methodology being implemented in any laboratory, including manufacturing plants, as a process analytical technology (PAT) tool in the pharmaceutical industry. 
We have tested the method with three fluorinated drug products available on the market - cinacalcet, lansoprazole, and ciprofloxacin - with doses ranging from 15 to 500 mg. The advantages of measuring the active content of fluorinated drugs using low-field 19F TD-NMR versus high-field 19F solid-state (SS) NMR are the simplicity of the method the low cost and the non-destructive nature of the technique, with all samples recoverable in intact forms ( e.g., powders, tablets, and capsules), making this technique affordable for any laboratory. This method is specific to fluorinated drugs because it detects only the content of fluorine, avoiding interference from the excipients that lack fluorine. 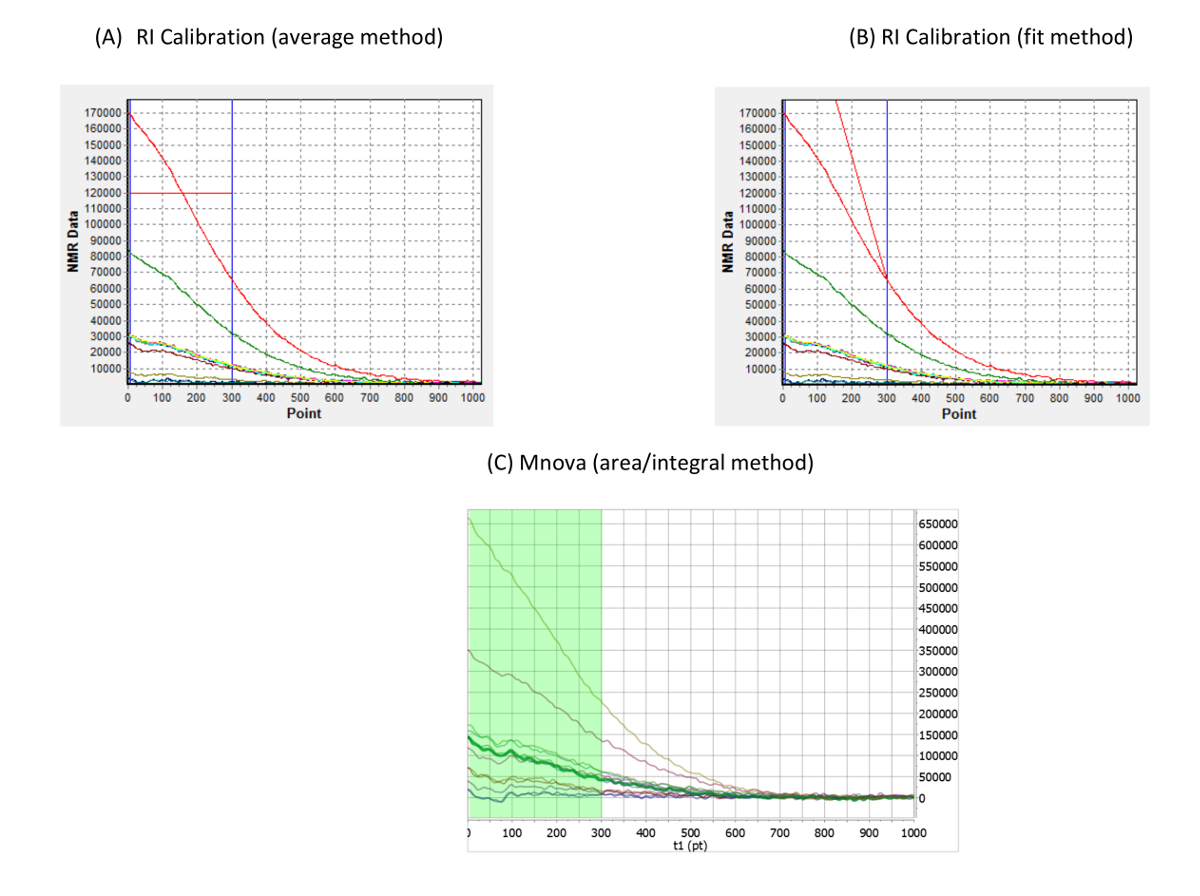
Here, we describe a protocol developed by our group that uses low-field fluorine-19 ( 19F) time-domain (TD) nuclear magnetic resonance (NMR) to measure the average content of fluorinated drugs in their formulated drug product forms: tablets or capsules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
